What is inflammatory arthritis?
Inflammatory arthritis (IA) is a general term utilized to characterize a group of diseases wherein the body’s immune system attacks the joints. The most often affected joints include the knees, hips, shoulders, and even fingers and toes. The most commonly recognized types of IA include: septic arthritis, gout, pseudogout, as well as autoimmune mediated inflammatory arthritis. The exact reason for someone to develop IA can vary, but ultimately, the body’s immune system goes into overdrive either due to a reaction to foreign organism (bacterial infection), reaction to crystal deposition inside the joint (gout and pseudogout), or the body’s own immune system incorrectly attacking itself (autoimmune mediated IA). In all cases, the joint becomes severely inflamed and can result in cartilage destruction, swelling, pain, and eventually destruction of the joint (arthritis).
Print Friendly
Print a copy of this article.
What is septic arthritis?
Septic arthritis, also known as infectious arthritis, occurs when bacteria enter the joint. The hip and knee joints are normally sterile and when bacteria enter, the bacteria itself, as well as the body’s own immune response to destroy the bacteria can damage the lining of the joint including the cartilage and bone surface. An organism can enter the joint from the bloodstream or through direct trauma. The most identified bacteria are Staphylococcus aureus, but other organisms have been found to be closely associated with certain diseases. For example, Salmonella is often seen in sickle cell disease Sickle Cell Anemia and Total Hip Replacement | Hip and Knee Care (aahks.org), and Candida albicans (a fungal infection) can be seen in patients who are immunocompromised (immune system is unable to fight infection). Most often, there is only a single organism that infects the joint, but in cases of a traumatic injury where the joint is penetrated with a foreign object or open cut to the joint, multiple bacteria types can be the culprit.
Patients will often present with severe pain, joint swelling, fevers (>101° F or 38.5° C) and will be unable to walk on the affected leg. In the hospital, the medical team will often place a needle in the joint to sample joint fluid (also known as a joint aspiration), which is then analyzed in the laboratory for detection of infection. (See Figure 1) The doctor will then use a combination of a patient’s symptoms and laboratory tests that help confirm a diagnosis of septic arthritis. This diagnosis is considered a surgical emergency (as soon as medically possible) and is usually treated surgically to wash out the bacteria from the infected joint. Additionally, patients are typically treated with several weeks to months of intravenous antibiotics to help minimize the chance of the infection coming back and to help return patients to their normal, active lifestyle. Sometimes, however, the infection results in cartilage destruction which can lead to severe arthritis.

What are gout and pseudogout?
Gout and pseudogout are two related types of IA conditions, which are also classified as crystalline arthropathies. In these two conditions, the body forms microscopic crystals inside the joint, which can result in an exaggerated immune response and swelling and pain in the joint. The most common locations for an attack are the big toe or the knee. Patients with a history of gout may be prone to repeat attacks throughout their lives and bouts most often occur after eating red meat, scallops/certain seafood, and drinking beer/alcohol. These conditions are diagnosed based on symptoms, and your doctor will be able to distinguish gout or pseudogout by sampling your joint fluid and analyzing the sample under the microscope. In gout, blood tests will often show high uric acid levels and the joint fluid analysis will show monosodium urate crystals, while pseudogout joint fluid samples show calcium pyrophosphate dihydrate crystals. The long-term consequence of untreated gout and/or pseudogout is potentially cartilage joint destruction. Treatment for both is similar and initially focuses on pain control with anti-inflammatory medications to treat the pain and swelling followed by maintenance medications that work to help the body eliminate these crystal byproducts. Gout is often treated with a non-steroidal anti-inflammatory medication such as colchicine initially (in severe situations oral or intravenous steroids may be given), and then allopurinol or other maintenance medications for chronic care to avoid recurrence. Severe cases of pseudogout may be treated with a steroid injection into the affected joint.
What is autoimmune-mediated inflammatory arthritis?
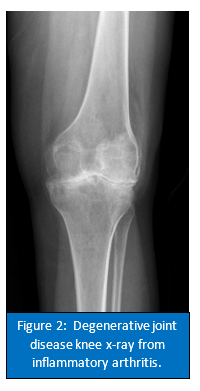
Autoimmune mediated inflammatory arthritis is a category of disease where the body’s own immune system attacks normal body cells and tissues and the immune system malfunctions and recognizes normal body tissue as a foreign invader, such as bacteria. This category of disease includes rheumatoid arthritis, juvenile idiopathic arthritis, seronegative spondyloarthropathy (for example: psoriatic arthritis, ankylosing spondylitis, reactive arthritis, arthritis associated with inflammatory bowel disease), and many other diseases. Unlike in septic or crystalline arthritis where foreign objects (bacteria or crystals) trigger an inflammatory response, in autoimmune mediated IA, the body’s own immune system inappropriately attacks the bone, cartilage, and surrounding soft tissues. This causes pain and swelling of the joint and if untreated can lead to joint degeneration (see Figure 2). The specific underlying condition can be differentiated by your doctor based on patient symptoms and specific lab values. Treatment is focused on decreasing symptoms of pain and swelling with anti-inflammatory medications, but long-term treatment involves special medications that directly alter the body’s inflammatory response. Commonly known medications to treat this condition include glucocorticoids (steroids), and the disease-modifying antirheumatic drugs (DMARDs) such as methotrexate, sulfasalazine, leflunomide, hydroxychloroquine. Biologic DMARD medications include etanercept, adalimumab, tocilizumab, infliximab, golimumab, abatacept, tofacitinib, among others. These medications have been a game-changer and have resulted in a more than 75% reduction in need for joint replacement surgery in patients with autoimmune mediated IA. However, these medications work by suppressing the immune system, which can also make the person taking the medication more susceptible to infections.
Total knee arthroplasty (TKA) Total Knee Replacement | Hip and Knee Care (aahks.org) and total hip arthroplasty (THA) Total Hip Replacement | Hip and Knee Care (aahks.org) are excellent options for patients with IA who have failed to improve with non-surgical management. Patients who should consider joint replacement are those who have had substantial joint space narrowing and likely bony destruction due to their inflammatory pathology, which causes them substantial pain and makes it difficult for them to ambulate. While THA and TKA are highly successful, patients with IA may be at increased risk of a complication, such as a postoperative infection. Particularly in patients with weakened immune systems, due to genetics or secondary effects from medications like DMARDs, the risk of infection following surgery can be higher when compared to those without altered immune systems. In other words, the advantage of DMARDs is to lower the body’s immune response to prevent it from inappropriately attacking itself, but by doing so, the body also has a blunted (or weaker) response for when it needs to battle infections. Post-operative infections can potentially require one, if not more, revision surgeries to help clear out the underlying infection Infection and Your Joint Replacement | Hip and Knee Care (aahks.org). In joint replacement surgery, multiple surgeries on the same joint have been associated with increased postoperative complications.
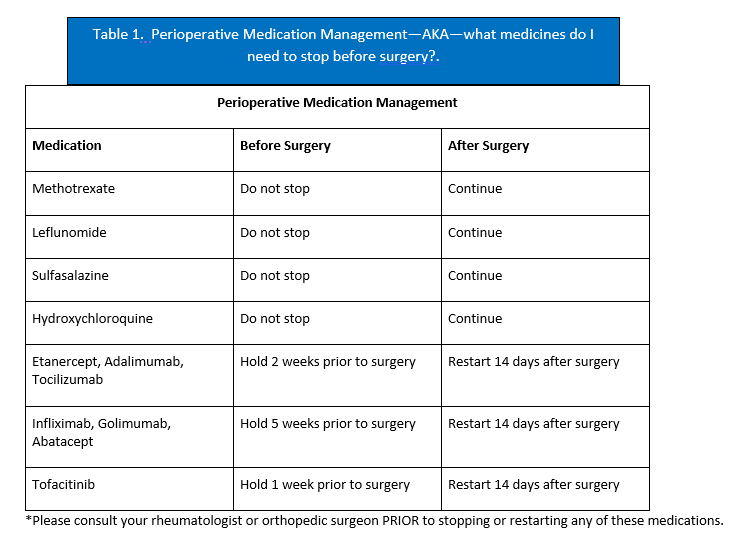
Despite the potentially increased infection risks, patients with IA should not be deterred from undergoing TKA, if otherwise appropriately indicated. Even patients who are taking DMARDs can still have successful outcomes. The most important steps are to work with your primary care, rheumatologist, and orthopaedic surgeon to make a surgical plan that accounts for any medications that you are taking. The latest recommendations from the American College of Rheumatology and the American Association of Hip and Knee Surgeons are that some DMARDs can be continued from before through after surgery, while others should be temporarily held before the operation and restarted at a particular schedule (Table 1). In any case, patients with IA should understand that there is an approximately 3x increased risk of complications, such as delayed wound healing and superficial or deep joint infection following joint replacement, but that risk can be lowered with appropriate preoperative optimization, medication management, and coordination amongst doctors.
Summary
If you suffer from any of these inflammatory arthropathies (autoimmune, gout, pseudogout, septic arthritis), contact your orthopedic surgeon for assistance with diagnosis and treatment. Many of these conditions can be managed safely, although special attention and additional communication will be critical if undergoing surgery such as a hip or knee replacement if you are on immune suppressive medications.
This article has been written by Jonathan R. Danoff, MD, in collaboration with the AAHKS Patient and Public Relations Committee and peer reviewed by the AAHKS Evidence Based Medicine Committee. Links to these pages or content used from the articles must be given proper citation to the American Association of Hip and Knee Surgeons.


